Among groups working on manual CHO cell banking workflows, two recurring priorities frequently surface:
minimizing DMSO handling
avoiding additional washing steps after thawing
These conversations reflect a broader industry shift toward safer, cleaner, and more operationally efficient cell banking processes.
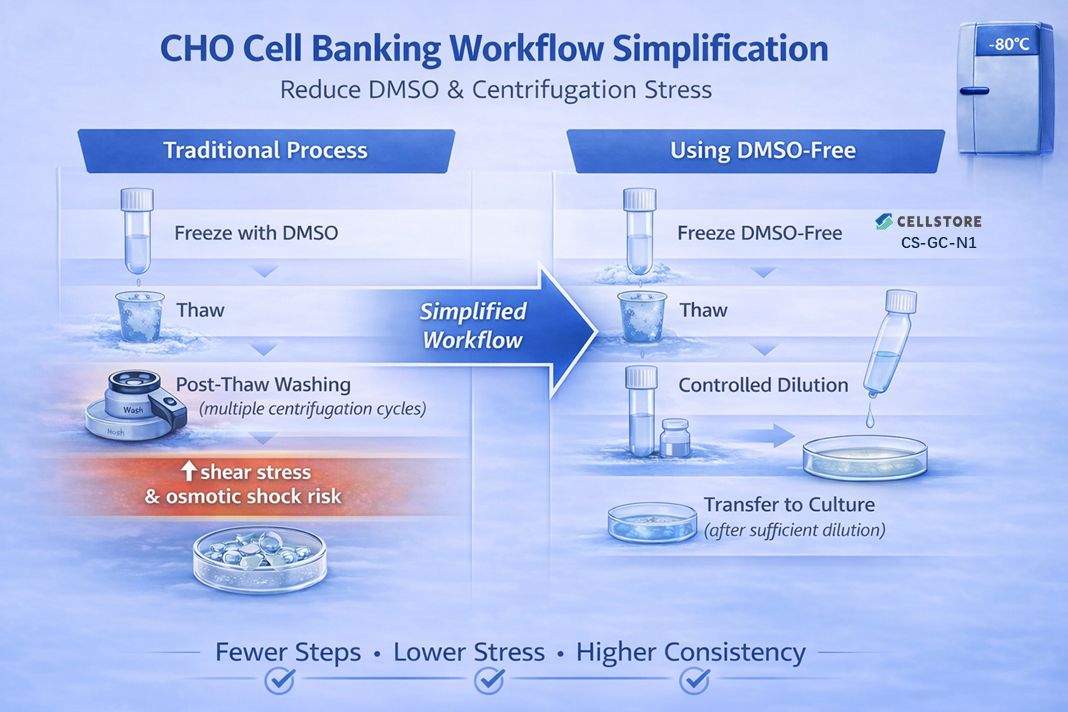
This is where CS-GC-N1 (DMSO-free cell line cryopreservation) becomes particularly relevant. Removing DMSO reduces toxicity concerns, and under sufficient dilution conditions, it may potentially support a more streamlined post-thaw workflow.
One aspect that often gets overlooked is the mechanical stress caused by centrifugation. Repeated spin–wash cycles can increase shear stress and osmotic shock, especially for sensitive CHO clones — and this is frequently where viability and recovery variability begin to emerge. A workflow that reduces or eliminates these centrifugation steps naturally lowers this risk.
It’s also important to consider osmolarity: cryopreservation media are naturally hyperosmotic compared to culture conditions, which is why washing is often included in standard SOPs. However, controlled dilution during thawing can help mitigate this impact depending on process setup.
We continue encouraging teams to run small, CHO-specific side-by-side evaluations — real workflows vary, and data always tell the truth.
As more biologics teams prioritize workflow simplification, DMSO-free solutions will play an increasingly meaningful role in future cell banking strategies.