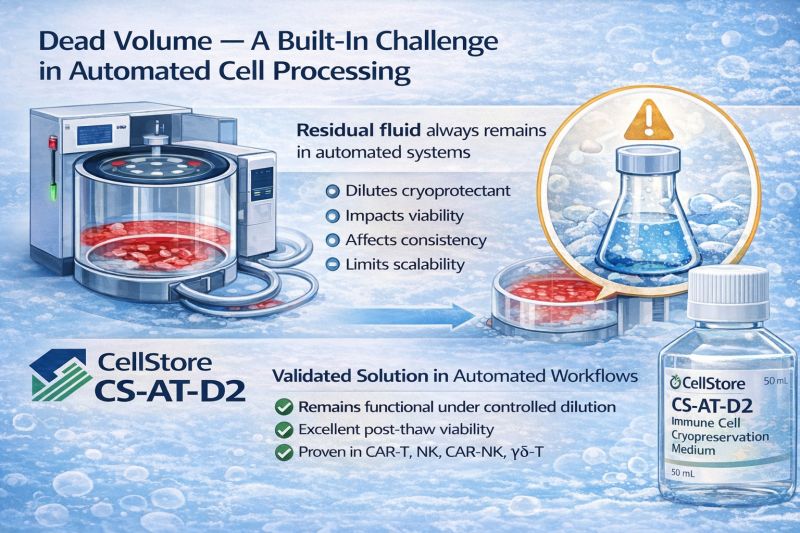
Dead volume isn’t a “bug” — it’s the reality of every automated cell processing system we use in CAR-T/NK manufacturing.
Whether your team runs Cytiva LOVO / Sefia, Thermo CTS Rotea, Miltenyi CliniMACS Prodigy, Lonza Cocoon, Fresenius Lovo/Amicus, or other continuous-flow centrifugation platforms, a small amount of residual fluid will always remain in the bowl or tubing.
This residual volume can dilute the cryoprotectant and influence:
post-thaw recovery
viability & osmolarity
batch consistency
downstream scalability
Most teams still choose between accepting dilution or adding extra formulation steps — neither ideal for scale-up.
In recent projects, we’ve been working with our industrial partners to identify cryoprotectant strategies that remain functional even under controlled dilution, so automation teams don’t need to redesign their entire Harvest process just to accommodate dead volume.
CS-AT-D2 is one solution we’ve repeatedly validated in automated workflows, and its consistency across CAR-T, NK, CAR-NK, and γδ-T processes has been strong.
If your team uses automated platforms, I’m curious how you handle residual volume across different systems — always happy to exchange notes.contact us to get more information.