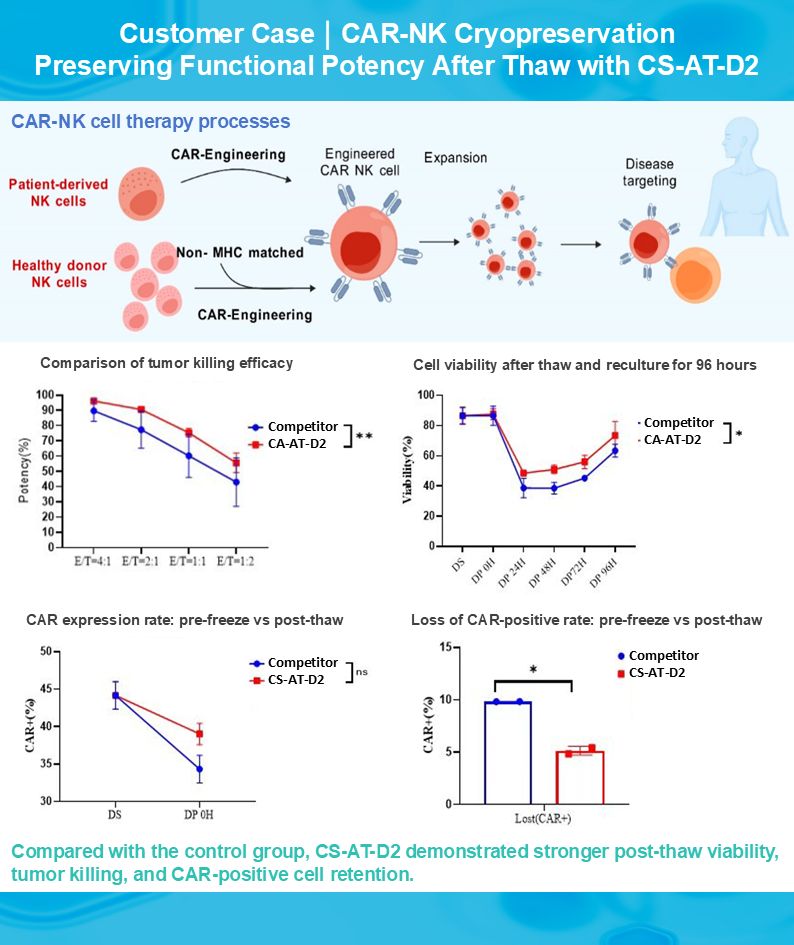
In CAR-NK cryopreservation, viability alone is not enough. Functional retention after thaw is what really matters.
We recently reviewed a CAR-NK cryopreservation validation case with CS-AT-D2, our cryopreservation medium designed for immune cells including T cells, NK cells, CAR-T, CAR-NK, αβ-T, and γδ-T cells, where the data showed encouraging post-thaw performance across several parameters relevant to downstream application, including:
☑️ sustained viability after thaw and reculture
☑️ stronger tumor killing efficacy versus comparator
☑️ better retention of CAR expression
☑️ lower loss of CAR-positive cells after freezing and thawing For engineered immune cell workflows, cryopreservation is not only a storage step. It is closely linked to consistency, potency, and translational value.
CS-AT-D2 has also been filed with the U.S. FDA DMF, supporting its relevance for advanced therapy workflows with higher requirements for quality and documentation.
As cell therapy continues to advance, evaluating cryopreservation media through both viability and functional performance will become increasingly important.