In industrial MSC manufacturing, the real challenge is often not freezing itself, but the waiting before it.
Between cell harvest, counting, resuspension, aliquoting, and freezing preparation, cells may remain in cryopreservation-related conditions longer than planned. In real workflows, this is not an exception — it is part of the process.
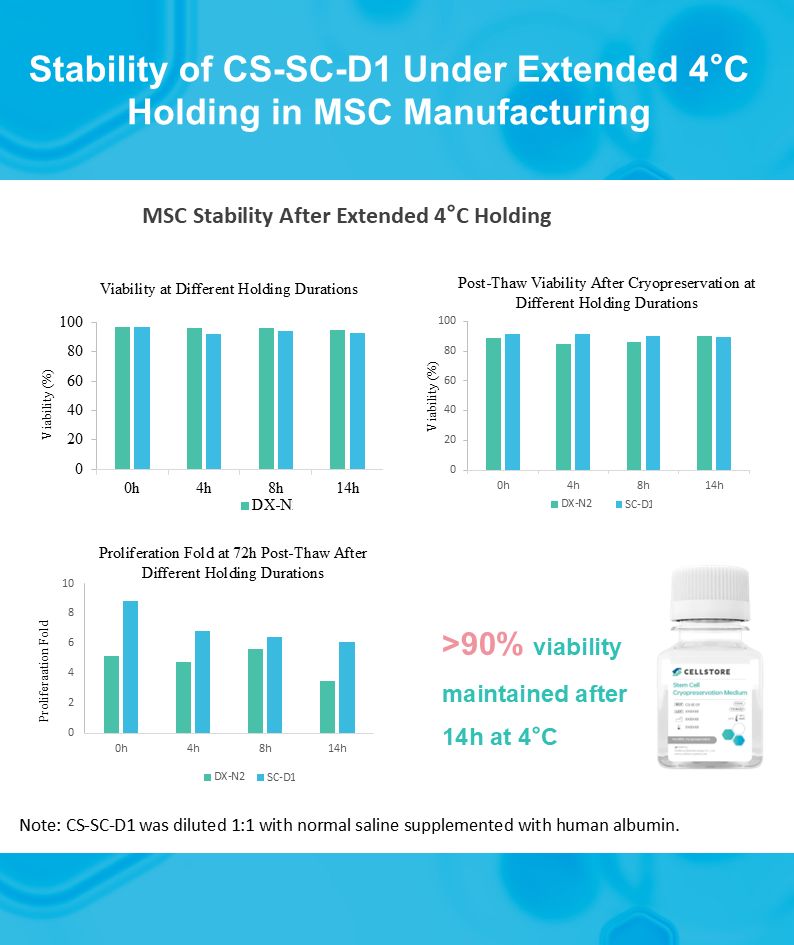
This raises an important question: how stable are cells during extended upstream holding?
To better reflect this scenario, we evaluated the stability of CS-SC-D1 Stem Cell Cryopreservation Medium under extended 4°C holding conditions.
CS-SC-D1 was diluted 1:1 with saline supplemented with human albumin and held at 4°C for up to 14 hours, simulating upstream temporary holding conditions in MSC manufacturing workflows.
What we observed:
✔️ Immediate viability remained above 90%
✔️ Post-thaw viability stayed around 90%
✔️ 72h post-thaw proliferation capacity was well maintained
In industrial production, processes rarely follow a perfect timeline. A cryopreservation medium that can tolerate real delays helps reduce operational pressure and improves workflow flexibility.
For MSC manufacturing, upstream stability is not just a technical detail — it is part of process robustness.